Information on treated water and decommissioning
(2024)
QDoes tritium affect our bodies?
ATritium emits weak beta rays (electrons). Exposure from the outside of our bodies (external exposure) is negligible. Exposure when taken into the body (internal exposure) is minimal.
Radiation emitted by tritium is weak beta rays (electrons). They can travel only up to 5 mm in the air, and exposure from the outside of our bodies is negligible.
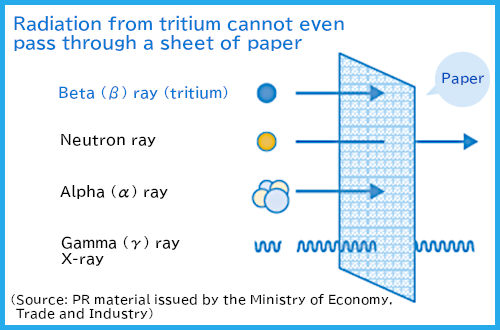
Figure 1 Radiation from tritium
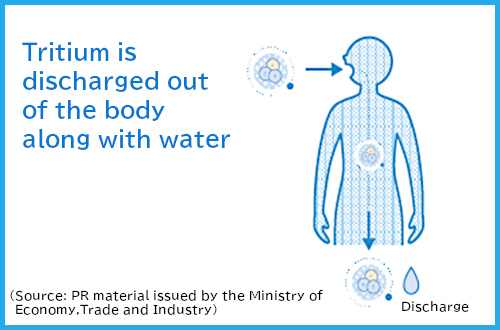
Figure 2 Internal exposure of tritium
The regulatory standard radioactivity concentration for tritiated water to be released as stipulated by the Japanese government is 60,000 Bq per liter. This value is set based on an estimate that the annual internal exposure will be 1 mSv when tritiated water with this radioactivity concentration is taken every day for 70 years.
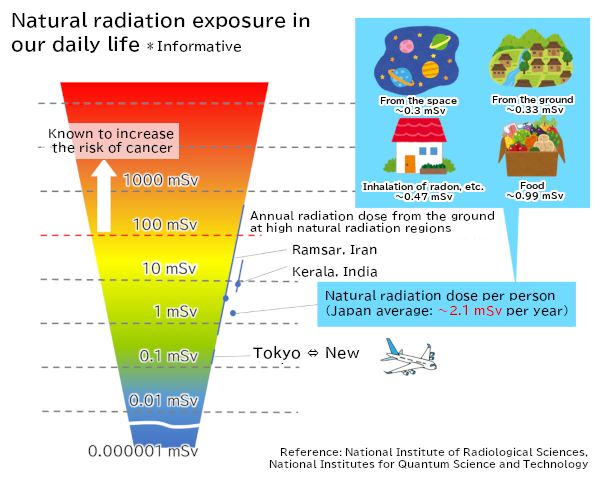
Figure 3 Exposure doses from natural radiations in our daily life
